|
Bio-organic
chemistry and Chemical biology
After the elucidation of the
genomes of several organisms the field of proteomics now faces the task
to
identify and characterize the functions of the genome products (i.e. the
expressed proteins in a cell,
tissue or organism). Most proteomic experiments deal with global
monitoring of
protein abundance, which is not necessarily related to function and
activity. Proteases are perhaps the best example of this discrepancy
between activity and abundance. Virtually all proteases are synthesized
as inactive zymogens and need to undergo an activation step. Once
active, their function is tightly regulated by post-translational
processes. To uncover the functions of proteases it is therefore
desirable to specifically monitor the dynamic levels
of their activities.
Chemical
or activity-based proteomics uses small molecule probes to specifically
modify small subsets of active enzymes. This allows to focus on the active subpartition of
proteins that are responsible for biochemical transformations.
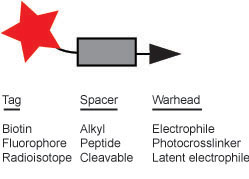
The
small molecules used in chemical proteomics are termed
activity-based probes (ABPs) and consist of three basic elements (see
Figure
1): a reactive warhead that is able to covelently modify an
enzyme’s active
site, a tag that facilitates enrichment and/or visualization of the
enzyme
activity, and a spacer that both separates the two previous elements
and
influences the selectivity of the reactive warhead.
One of the major research areas
of our laboratory is the development of new activity-based probes. We
make use of both solution and solid-phase chemistries
to create straightforward
syntheses that can easily be carried out and facilitate optimization
for
different protease targets. With these probes we aim to uncover
regulation mechanisms of protease activity and uncover proteases as
diagnostic markers and targets for therapeutic intervention.
|
|
News
For more information about the scientific work of Steven Verhelst, please visit the homepage of his lab.
|